RADWAG Balances and Scales (Poland) - XA 6.4Y.M.A PLUS STENT Balance in the Hands of Biomer Technology, a British Company.
Biomer Technology is a UK-based company that specialises in the design and manufacture of polymers for challenging applications in both the medical device and specialist industrial markets. Biomer Technology is based in Runcorn in the North West of England.
RADWAG lent the XA 6.4Y.M.A PLUS STENT microbalance to Biomer for testing. Before we move forward to the results and conclusions, let us explain what a stent is. A stent is a type of vascular prosthesis that is placed inside a blood vessel to restore its patency. It has a cylindrical shape and a fine mesh structure. Stents are used in cases of cardiovascular disease.
How does Biomer Technology summarise months of work with the XA 6.4Y.M.A PLUS STENT microbalance manufactured by RADWAG? We asked Simon Dixon, a Biomer representative, about this.
He said:
– Understanding the many challenges faced when applying thin, polymeric coatings to complex geometry devices such as stents is critical. This is further complicated by the lack of commercially available non-destructive methods to accurately quantify the amount and distribution of coating applied, both in process and post processing.
The thin layer coatings, by their very nature, are difficult to assess optically, and in the case of a semi- or transparent coating applied at single digit micron level, it is not always possible to even determine if the coating process has been successful. Mass determination, however, allows for rapid, in-process, non-destructive inspection of products, and in the manufacturing environment provides a reproducible quality control check.
Biomer Technology has been using weight determination as part of its stent coating research and for in-process quality control checks for many years. Weight determination offers the immediate, non-destructive and reproducible feedback that is lacking in many other analytical techniques used in coating development.
This is not to say that weight determination can provide a stand-alone result, as one still requires a parallel optical verification technique to determine coating quality and distribution. It is also not without its own challenges, primarily with device handling, and it is in this regard that I believe RADWAG’s novel stent toolkit has made a considerable leap forward.
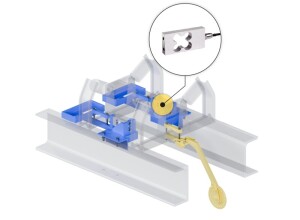
The toolkit replaces the traditional small pan systems with a frame and applicator, allowing the operator to quickly (and reproducibly) load, measure and unload the device. In the research environment, combined with appropriate optical characterisation means, it can help reduce the iterative development cycle by providing in-process feedback allowing for rapid optimisation of coating weight. The ability to approximate coating thickness from weight and distribution during process development can significantly lower development time and the additional costs of off-line destructive testing techniques. The process is especially beneficial where multilayer coatings are required, either to achieve a specific thickness or as differential layers in the case of active coatings containing API’s.
Having worked with the RADWAG prototype system for several months now, I believe it represents a significant opportunity for both developers and manufacturers of coated stent systems to improve the efficiency and quality of the products and services that they provide – Mr Simon Dixon concluded.
We are glad to know that XA 6.4Y.M.A PLUS STENT microbalance of RADWAG production meets the expectations of Biomer Technology and we hope for a fruitful cooperation in the future.















Interested? Submit your enquiry using the form below:
Only available for registered users. Sign In to your account or register here.